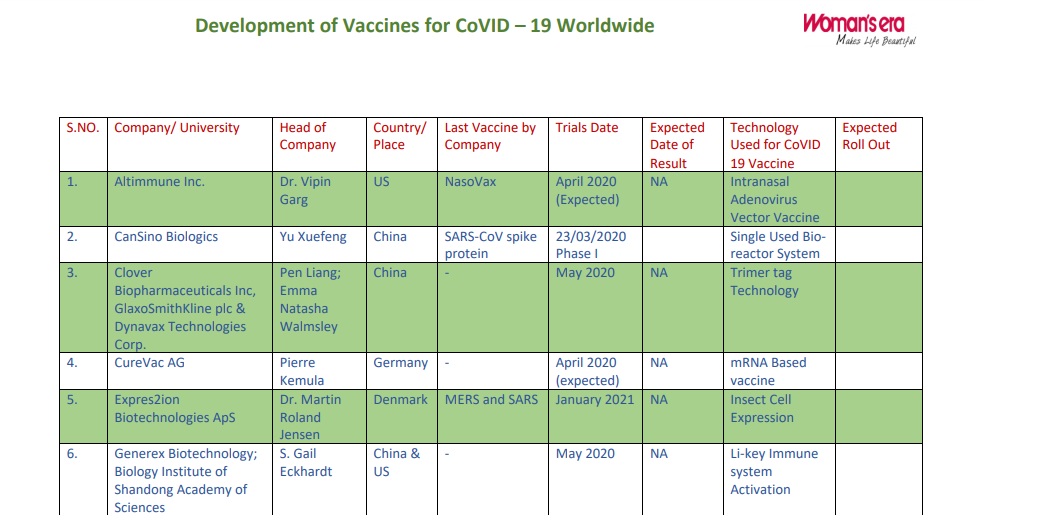
| S.NO. | Company/ University | Head of Company | Country/ Place | Last Vaccine by Company | Trials Date | Expected Date of Result | Technology Used for CoVID 19 Vaccine | Expected Roll Out |
| 1. | Altimmune Inc. | Dr. Vipin Garg | US | NasoVax | April 2020 (Expected) |
NA | Intranasal Adenovirus Vector Vaccine | |
| 2. | CanSino Biologics | Yu Xuefeng | China | SARS-CoV spike protein | 23/03/2020 Phase I |
Single Used Bio-reactor System | ||
| 3. | Clover Biopharmaceuticals Inc, GlaxoSmithKline plc & Dynavax Technologies Corp. | Pen Liang; Emma Natasha Walmsley and CEPI | China | – | May 2020 | NA | Trimer tag Technology | |
| 4. | CureVac AG | Pierre Kemula and CEPI | Germany | – | April 2020 (expected) |
NA | mRNA Based vaccine | |
| 5. | Expres2ion Biotechnologies ApS | Dr. Martin Roland Jensen | Denmark | MERS and SARS | January 2021 | NA | Insect Cell Expression | |
| 6. | Generex Biotechnology; Biology Institute of Shandong Academy of Sciences | S. Gail Eckhardt and Melinda Foundation | China & US | – | May 2020 | NA | Li-key Immune system Activation | |
| 7. | Ibio Inc; Beijing CC Pharming Ltd. | Robert B. Kay | China | – | Approval waited for clinical trials | NA | FastPharming Sytem (Plant Produced) | |
| 8. | Imperial College of London | Robin Shattock and CEPI | UK | – | April 2020 (expected) |
NA | Antibody therapy | |
| 9. | Inovio Pharma; Beijing Advaccine Biotech. Co. Ltd; Genome Lifescience Inc. | J. Joseph Kim and Funds By Bill Gates | China & US | MERS | April 2020 (Human trials expected) |
September 2020 | DNA Vaccine | |
| 10. | Linearx Inc; Takis Biotech | James Hayward | New York & Italy | April 2020 (expected) |
NA | Polymerase Chain Reaction | ||
| 11. | Medicago Inc. | Dr. Bruce Clark | Canada | SARS CoV 2 gene | July 2020 (expected) |
NA | Basic Antibodies response | |
| 12. | Moderna Therapeutics Inc.; Kaiser Permanente Health Research Inc.; NIAID | Bill Gates and CEPI | US | Zika Vaccine, SARS 1 | 16/03/2020 (Human trial) |
NA | mRNA based Vaccine | |
| 13. | Novavax Inc. | Staney C. Erck and CEPI | US | MERS | May 2020 (expected) |
NA | Nanoparticles vaccine | |
| 14. | The Jenner Institute; University of Oxford | Prof. Adrian Hill | UK | MERS | 28/03/2020 (Phase I) |
NA | Adenovirus Vector Encoding | |
| 15. | Stermirna therapeutics Co. Ltd.; Tongji University | Li Hangwen | China | – | April 2020 (expected) |
NA | mRNA based vaccine | |
| 16. | University of Queensland | Peter Hoj and CEPI | Australia | – | May 2020 (expected) |
NA | Protein subunit based | |
| 17. | University of Saskatchewan | Richard Peter Stoicheff | Canada | – | NA | NA | Different strains of coronavirus | |
| 18. | Vaxart Inc. | Wouter W. Latour | UK | – | June 2020 (expected) |
NA | Recombinant protein vaccine | |
| 19. | Vaxil Bio Ltd. | Ari S. Kellen | Israel | – | July 2020 (expected) |
NA | Peptide vaccine based on bioinformatics | |
| 20. | Walter Reed Army Institute of Research and US Army Medical Research and Development Command | Col. Deydre S. Teyhen | US | Ebola | July 2020 (expected) |
NA | MERS research | |
| 21. | Zydus Cadilla | Pankaj Patel | India | – | NA | NA | rMeasles Virus vector based | |
| 22. | Mayo Vaccine Research Group | Dr. Gregory Poland | China | – | 25/03/2020 | NA | Based on Peptide Subunit | |
| 23. | University of Western Ontario | – | Canada | – | May 2020 (expected) |
NA | Bioinformatics and genetic imaging | |
| 24. | National School of Tropical Medicine | Peter Hotez | US | SARS 1 | May 2020 (expected) |
NA | Development from SARS | |
| 25. | Cambridge University | Jonathan Henley | UK | Ebola | Looking for Funds | NA | Antibodies induced enhancing | |
| 26. | State Key Laboratory for Emerging Infectious Diseases | Xiang Zhang | Hong Kong, China | H1N1 | April 2020 | NA | Based on Flu Vaccine | |
| 27. | BioNTech SE and Pfizer Inc. | Ugur Sahin | Germany | Cancer related | April 2020 (Expected) |
NA | mRNA baaed | |
| 28. | CytoDyn Inc. | Nader Z. Pourhassan | USA | HIV | April 2020 (Expected) |
NA | Antibodies response based | |
| 29. | Heat Biologics Inc. | Jeffrey Wolf | USA | Immunotherapy | April 2020 (Expected) |
NA | Based on Flu Vaccine | |
| 30. | Johnson & Johnson | Alex Gorsky | USA | HIV | September 2020 | NA | mRNA Based | |
| 31. | Regeneron Pharmaceuticals Inc | Leonard Schleifer | USA | Ebola | May 2020 | NA | Peptide bond Based | |
| 32. | Sanofi | Paul Hudson | France | Rubella | April 2020 (Expected) |
NA | – | |
| 33. | India’s Serum Institute | Cyrus Poonawalla | India | Swine Fly | April 2020 (Expected) |
January 2021 | Antibodies response Based | |
| 34. | Bharat Biotech | Dr. Krishna Ella | India | RotaVac | April 2020 (Expected) |
December 2020 | Nasal Vaccine | |
| 35. | GeoVax and BravoVax | Farshad Guirakhkoo | US | HIV | NA | NA | – | |
| 36. | University of Pittsburgh | Louis Falo | US | MERS | 2 April 2020 Preclinical Trial |
NA | Based on SARS CoV 1 Vaccine |
Updated on: 05/04/2020
Created on: 29/03/2020
Compiled by: Himshikha Shukla
Editor: Divesh Nath